Leukopaks, Non-Mobilized
Access Fresh And Cryopreserved Leukopaks From Healthy And Diseased Donors For Research
Gain access to a growing network of providers for fresh and cryopreserved leukopaks from a large collective donor pool and diversity of phenotypes.
Select from a list of donor attributes such as blood type, HLA type, body mass index (BMI), gender, age, race, ethnicity, smoking (tobacco, vape or THC), or simply choose a standard Leukopak. You also have the ability to specify annotation requirements and exclusion criteria.
Each Donor is assessed for medication, lifestyle, and medical history, tested for HBV, HCV, HIV and serological tests as a minimum, with an option to test for an expanded panel of infectious diseases.
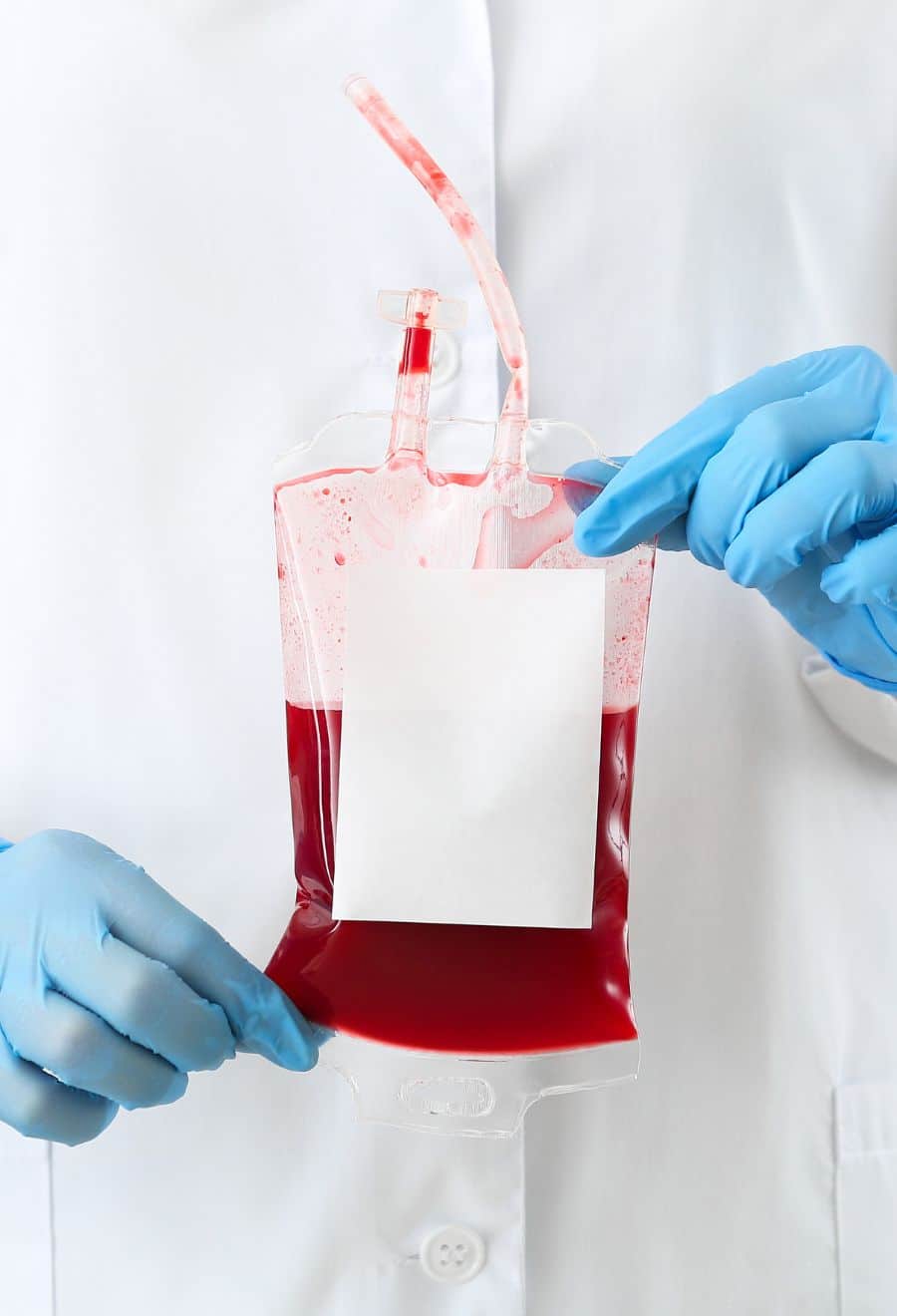
The iSpecimen Advantage When Sourcing Leukopaks for Research
Leukopaks are concentrated, low-volume apheresis collections from normal donors, approved by IRB or REC protocols.
iSpecimen simplifies the process of sourcing reliable, highly-quality leukopaks while also offering many of the following benefits:
Specificity of Donor Attributes
Refine inclusion and exclusion criteria from a wide range of attributes, including age, gender, race, ethnicity, blood type, smoker status, and more.
Diseased Mononuclear Cells
iSpecimen also offers diseased mononuclear cells through its continually growing network of partners.
Optimized Cellularity & Cell Counts
Available fresh or frozen, our leukopak collections typically yield over 10 billion WBCs within a sample volume of 300–500 ml.
Mobilized & Non-Mobilized Leukopaks
Depending on your research and cell count requirements, choose from mobilized or non-mobilized leukopaks, all from your desired phenotype.
Cryopreserved, Healthy & Diagnosed Donors
In addition to fresh leukopak collections, our network offers access to a large inventory of healthy and diseased cryopreserved, mobilized leukopaks.
Compliance Assurance
Compliance is ensured. Providers are in conformance with U.S. CFR regulations under the oversight of an Independent Review Board (IRB).
Fresh and Cryopreserved Leukopaks, Detailed Information
iSpecimen's fresh and cryopreserved Leukopaks are provided for research and development use only, not for human therapeutic or diagnostic use.
Choosing the Best Leukopak Option for Your Research
The iSpecimen Marketplace provides access to fresh and frozen, healthy and diseased high-quality leukopaks, as defined by cell counts, percent cell viability, cellularity, validated packaging, and on-time delivery.
Collection lead times are typically two weeks for non-mobilized leukapheresis collections and six weeks for mobilized leukapheresis collections; lead times may vary depending on specificity of requested donor characteristics and collection criteria. Leukopaks can be shipped under cold or ambient conditions as requested, with an option to include a temperature logger.
Find a summary of options and additional details for each, below.
Fresh Non-Mobilized Leukopaks
- Fresh collection, healthy donors
- Full leukopaks, >300 ml
See full details below
Fresh Mobilized Leukopaks
- Fresh, healthy normal
- Max yield, >50 billion cell count
- Days 1-2-3-4-5 (second day collection available)
- Full >300 ml
Cryopreserved Leukopaks, Healthy & Diagnosed
- Healthy normal
- Diagnosed, Cancer
- Diagnosed, Autoimmune Diseases
- See full details below
Finding the Right Leukopak & Donor Match for Your Research
In the iSpecimen Marketplace®, researchers can request fresh or frozen mobilized and non-mobilized leukopaks from healthy and diagnosed patients fitting specific donor profiles, including but not limited to:
- Blood Type
- HLA Type
- Body Mass Index (BMI)
- Demographics (Gender, Age, Ethnicity, Race)
- Smoking Status (tobacco, vape or THC).
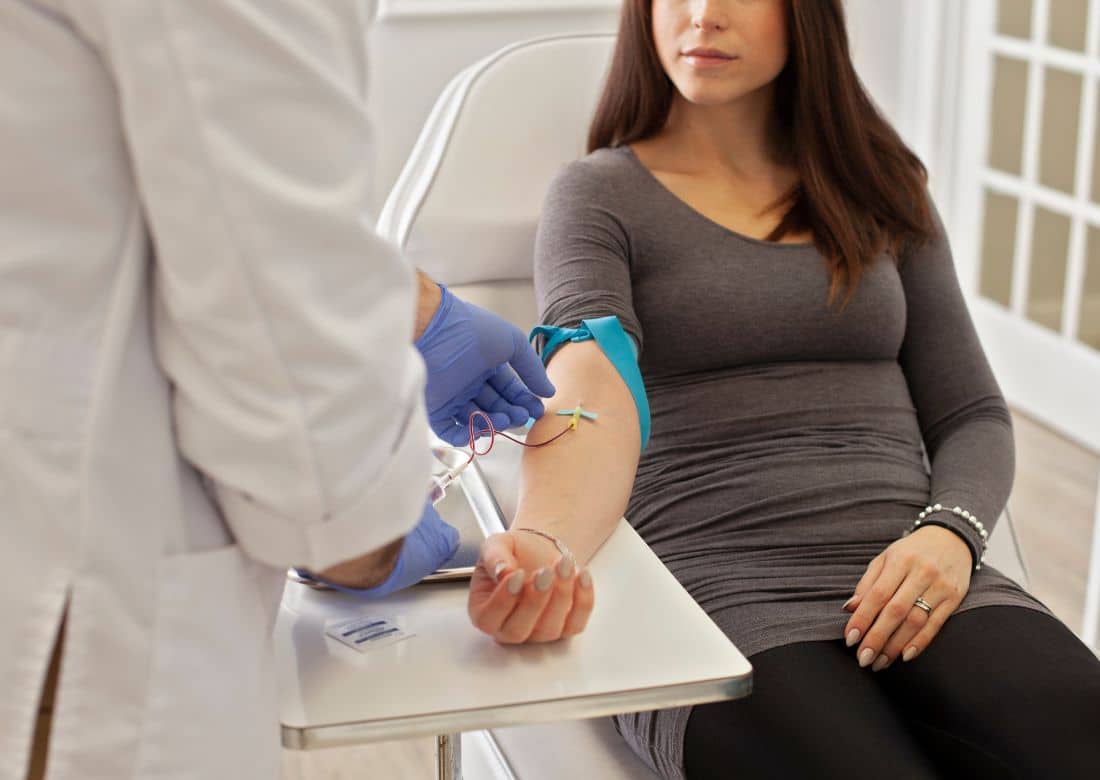
A New, Efficient Way of Sourcing Leukopaks
Overnight Leukopak Shipping & Delivery
Recently, we arranged for the collection and overnight delivery of fresh whole leukopaks from adult non-smoker to a research organization on the east coast. If you need highly-specific, readily-available leukopaks, iSpecimen can help.

The World is Your Biobank
Expand Your Patient Sets by Tapping into Our Global Network
Reach beyond restricted or limited patient populations to find the human tissue, blood products, and other biofluid specimens needed to accelerate your research.
Biospecimen Matching for Your Research
You need access to biospecimen samples that mirror your patient population's genetics and physical attributes. With iSpecimen, you have more opportunities to source specific, diverse samples.
Access the Leukopaks You Need for Your Research
FAQs
There are several differences between frozen tissue and FFPE tissue including cell concentration and usage:
Leukopaks are enriched with a high concentration of mononuclear cells (T cells, B cells, NK cells, and monocytes), and are preferred for cellular therapies, research requiring high cell yields, and specific immune cell isolation.
Whole blood contains all blood components (red cells, white cells, platelets, plasma) in normal physiological concentrations. This biofluid is often used for genetic work, validating medical instruments, or developing blood-based testing methods.
The yield is dependent on the type of leukopak, but typically yields over 10 billion WBCs within a sample volume of 300-500 ml with a minimum cell viability of >97%.
Yes, all samples are provided with relevant information including full product details (including cell yield and percent cell viability) as well as donor information such as patient demographics, diagnoses, medications, procedures, outcomes data, lab test results, pathology reports, family history, and social history.
Researchers can identify selectable attributes using a donor profile, including demographics, diagnoses, medications, etc. Donors are then recruited for collections according to these attributes ensuring that you receive the specific specimens needed for your research.
While we do not have standard volume deals for bulk orders, we are committed to exceptional customer service. We are open to discussing and implementing pricing adjustments or discounts based on your specific needs and order size.
Collection lead times are typically two weeks for non-mobilized leukapheresis collections and six weeks for mobilized leukapheresis collections, but may vary depending on the specificity of requested donor characteristics and collection criteria. Shipping times vary by location but are expedited due to sample preservation.
